Though we cannot be absolutely sure that the total entropy of the whole universe is increasing, we can say that, wherever it can be scientifically tested, the entropy in any given portion of the universe is increasing. However, wherever and whenever the entropy principle has been subjected to scientific test, it has always worked, with no exception. This stricture is philosophically valid, of course. Empirical measurements can never establish universal certainty. Since man is quite limited in knowledge, and since he is able to make observations on only a very small part of the vast universe, he cannot be certain that the Second Law applies everywhere in time and space.

One is to deny the universality of the entropy principle.

Creationists, therefore, insist that the evolutionary philosophy must simply be wrong.Įvolutionists point to two possible ways out of this difficulty. It is certainly not a scientific law, as is the Second Law of Thermodynamics, which it contradicts. It is not even a good scientific hypothesis, since there is no way to subject it to scientific test. No one knows how it works, or has ever seen it take place. Since the Second Law of Thermodynamics is universally accepted as a basic principle governing all processes, has been verified experimentally thousands of times, and is consistent with all experience, there can be no doubt whatever that, if there is such a thing as a scientific law at all, this is it! Evolution, on the other hand, is simply a belief. Thus, according to Huxley, evolution is a universal law requiring that all processes lead irreversibly toward an “increase of order.” According to Blum, the entropy principle is a universal law requiring that all processes lead irreversibly toward a “decrease of order.” Each is exactly the converse of the other! It seems obvious, therefore, that one of them must be wrong. The entropy also measures the randomness or lack of orderliness of the system, the greater the randomness the greater the entropy. All real processes go with an increase of entropy. Any given process in this universe is accompanied by a change in magnitude of a quantity called the entropy. The Second Law of Thermodynamics has as one of its consequences that all real processes go irreversibly. The outstanding Princeton bio-chemist, Harold Blum, describes this law in the following way:

The Second Law of Thermodynamics is also known as the Law of Increasing Entropy. Thus, evolution encompasses all reality: particles evolve into atoms and atoms into molecules and molecules into worlds and stars and galaxies inorganic compounds evolve into living materials and these into more and more complex plants and animals and finally into man, who now presumably can intelligently control all future evolution. Our present knowledge indeed forces us to view that the whole of reality is evolution-a single process of self-transformation.
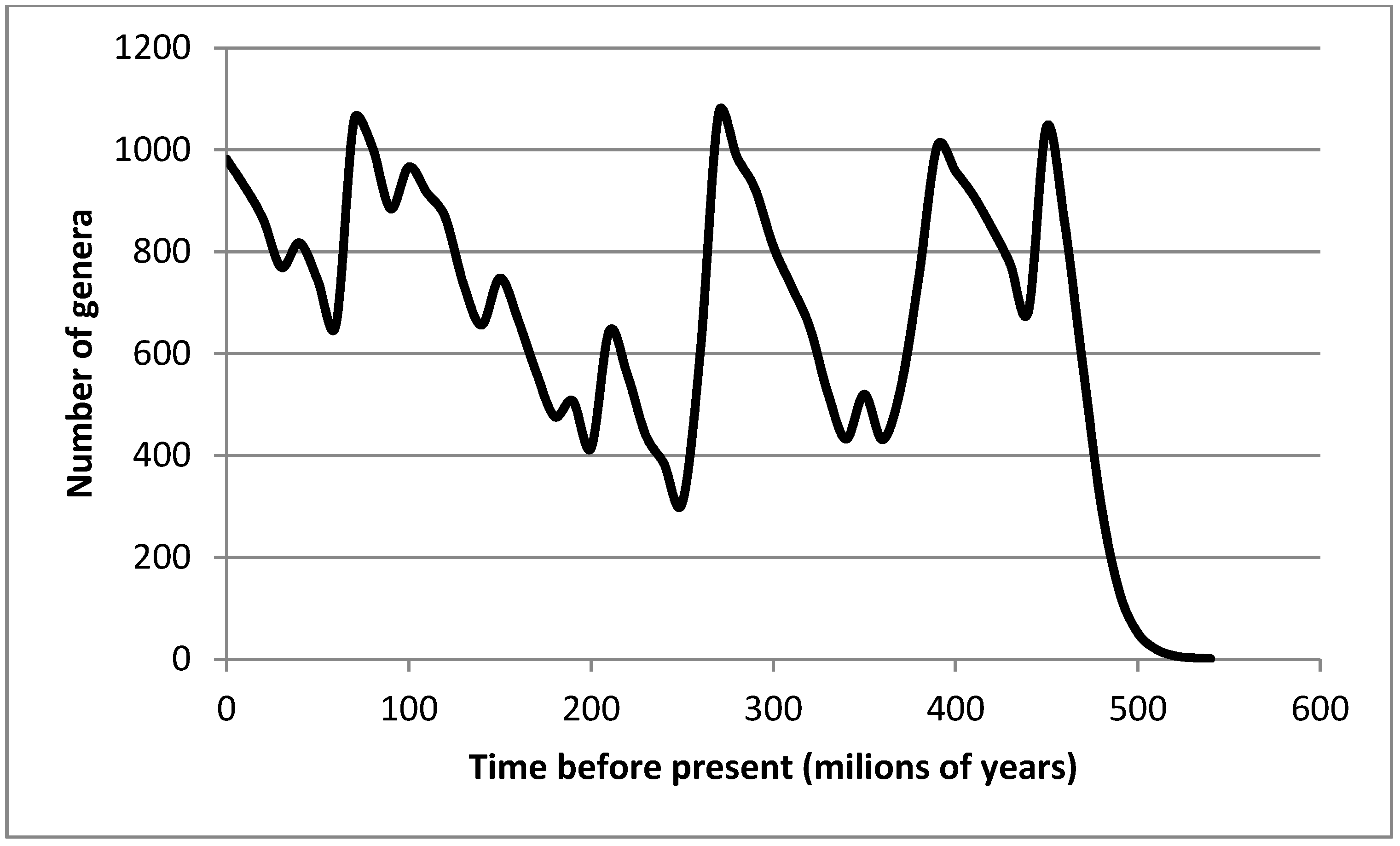
Sir Julian Huxley, probably the world’s greatest modern evolutionist, has defined evolution as follows:Įvolution in the extended sense can be defined as a directional and essentially irreversible process occurring in time, which in its course gives rise to an increase of variety and an increasingly high level of organization in its products. It is well to let two leading evolutionists define the two concepts. Evolutionists, however, sometimes argue that there is no contradiction and that both can be true. One of the arguments which creationists have used effectively against evolution is that the evolutionary hypothesis of the development of the cosmos and of the present organic world is contradicted by the entropy principle, the Second Law of Thermodynamics. Can the theory of evolution be harmonized with the Second Law of Thermodynamics?
